Specifically targeting and manipulating living cells is a key challenge in biomedicine and in cancer research in particular. Several studies have shown that nanoparticles irradiated by intense lasers are capable of conveying damage to nearby cells for various therapeutic and biological applications. In this work we utilize ultrashort laser pulses and gold nanospheres for the generation of localized, nanometric disruptions on the membranes of specifically targeted cells. The high structural stability of the nanospheres and the resonance pulse irradiation allow effective means for controlling the induced nanometric effects. The technique is demonstrated by inducing desired death mechanisms in epidermoid carcinoma and Burkitt lymphoma cells, and initiating efficient cell fusion between various cell types. Main advantages of the presented approach include low toxicity, high specificity and high flexibility in the regulation of cell damage and cell fusion, which would allow it to play an important role in various future clinical and scientific applications.
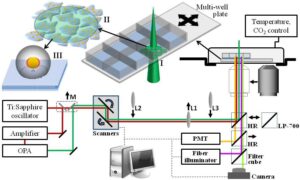
Figure 1: Schematic of the optical system for nano-manipulations of cells. A beam from an optical parametric amplifier (OPA) at wavelength 550 nm (green line) was scanning the sample using two scanning mirrors. A single lens (L1) was used to reduce the beam’s diameter on the sample to 250-350 micron. For two-photon imaging, the beam from the Ti:Sapphire oscillator was picked using a flipped mirror (M), scanned and magnified using lenses L2 and L3, and focused by inserting an objective lens before the sample. The two-photon fluorescence signal was detected by a photo-multiplier tube (PMT) after replacing the high reflectivity (HR) mirror by a dichroic short-pass mirror (LP-700). After irradiation, time lapse imaging was conducted by replacing the dichroic mirror with different filter cubes optimized for the specific fluorescence markers. Schematic illustrations show (I) the irradiation pattern of the pulse beam, (II) the irradiated nanoparticle-targeted cells, and (III) the extent of the nanometric effect induced by each nanoparticle, including the near field enhancement beyond the ionization threshold (red regions), the spherical shock wavefront (gray sphere), and the affected area on the membrane (dashed circle).
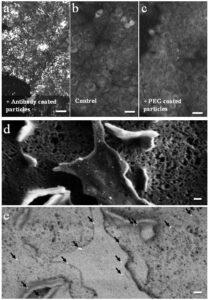
Figure 2: Gold nanoparticles on carcinoma cell membranes. Two-photon imaging of A431 cells incubated with (a) anti-EGFR coated gold nanoparticles, (b) no nanoparticles and (c) PEG-coated gold nanoparticles. Scale bars in a-c represent 30 micron. (d) Scanning electron microscopy of a cell membrane targeted by anti-EGFR gold nanoparticles. (e) Scanning electron microscopy using the back scattering detector reveals bright reflections indicative of gold particles (marked by arrows). Scale bars in d-e represent 100 nm.
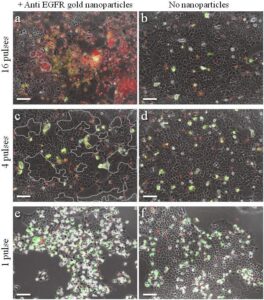
Figure 3: Damaging carcinoma cells using anti-EGFR coated nanoparticles and resonance femtosecond pulse irradiation. (a) Gold nanoparticle-conjugated cells irradiated by 16 pulses. (b) Non-conjugated cells, 16 pulses. (c) Conjugated cells irradiated by 4 pulses. (d) Non-conjugated cells, 4 pulses. (e) Conjugated cells irradiated by 1 pulse. (f) Non-conjugated cells, 1 pulse. Multi-nucleated cells margins are marked by white dashed curves in (c). Scale bars represent 50 micron. Red nuclei indicate necrotic cells. Green stain indicates apoptosis. Panels a-d show cells 5 hours after irradiation. Panels e, f show cells 23 hours after irradiation.
Figure 4: Induction of cell fusion. (a) Time sequence of fluorescence images of fusing BJAB cells superimposed on phase contrast images, following irradiation by 5 pulses in the presence of non-specific gold nanoparticles. Plasma membranes were green labeled. (b) Formation of a hybridoma cell. Time sequence of fluorescence images of human BJAB cells (red nuclei) and murine (NSO) cells (blue nuclei) superimposed on phase contrast images, following irradiation of 5 pulses in the presence of non-specific gold nanoparticles. Scale bars represent 10 micron.
References
- Optical Nano-Manipulations of Malignant Cells: Controlled Cell Damage and Fusion
Limor Minai, Daniella Yeheskely-Hayon, Lior Golan, Gili Bisker, Eldad J. Dann and Dvir Yelin
Small , 8, 1732 (2012) - High levels of reactive oxygen species in gold nanoparticle-targeted cancer cells following femtosecond pulse irradiation
Limor Minai, Daniella Yeheskely-Hayon and Dvir Yelin
Sci. Rep. 10.1038, srep02146 (2013) - Experimental proof for the role of nonlinear photoionization in plasmonic phototherapy
Limor Minai, Adel Zeidan, Daniella Yeheskely-Hayon, Shimon Yudovich, Inna Kviatkovsky, Dvir Yelin
Nano Lett. 16, 4601 (2016) - Plasmonic targeting of cancer cells in a three-dimensional natural hydrogel
Limor Minai, Matan Hamra, Dvir Yelin,
Nanoscale, 10, 17807 (2018)